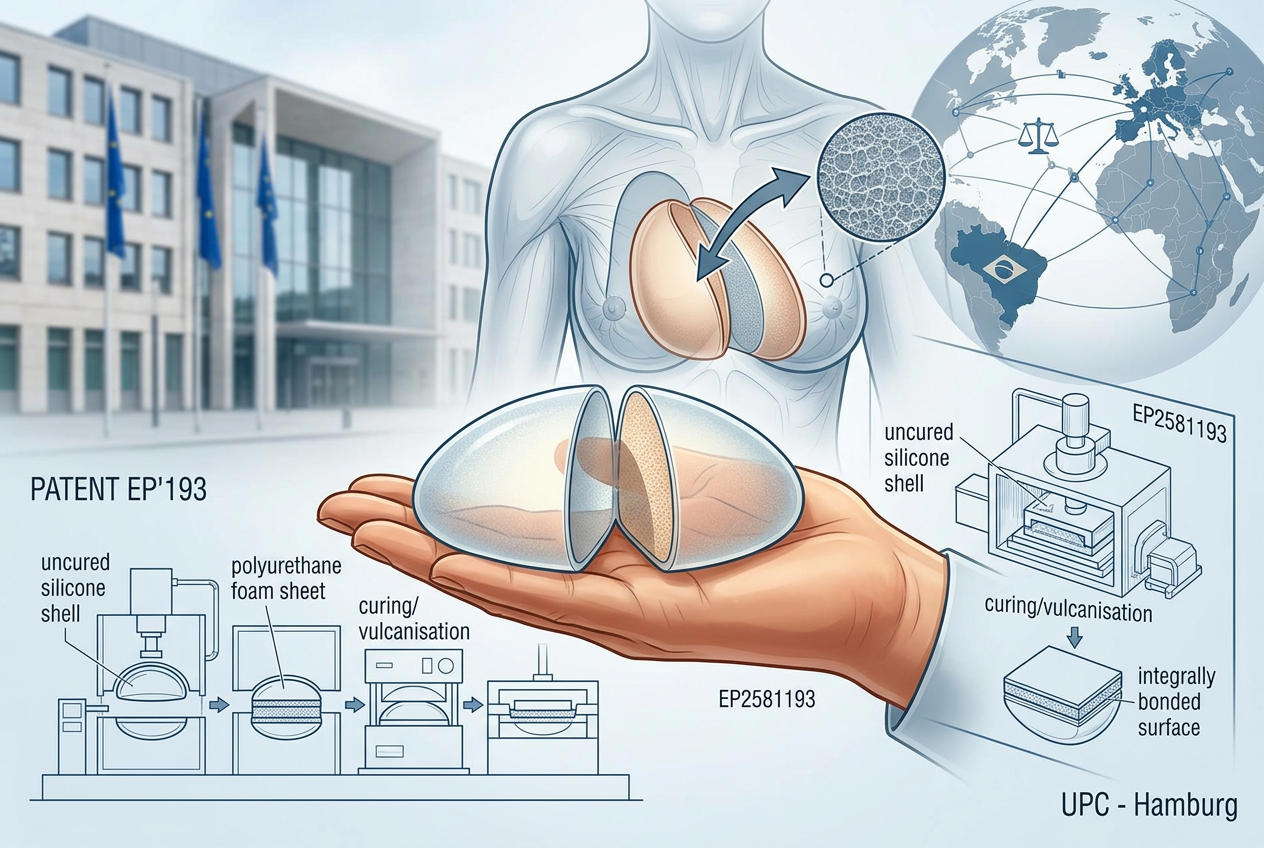
Brazilian medical device manufacturer Silimed Indústria de Implantes Ltda has filed an infringement action before the Unified Patent Court (UPC), Hamburg Local Division, becoming the first Brazilian company to initiate proceedings before the UPC.
The action targets Polytech Health & Aesthetics GmbH and several alleged European distributors and concerns European Patent EP 2 581 193 B1 (EP’193), which protects a process for manufacturing polyurethane-coated silicone implants, in particular breast implants.
Silimed’s filing marks a notable development for the UPC. Since the court opened in 2023, most claimants have been European, US or Asian companies. The Hamburg case is the first time a Brazilian company has turned to the court as a centralised enforcement forum.
EP’193 covers a multi-step manufacturing process involving, inter alia, (i) providing an implant shell comprising uncured silicone, (ii) placing a polyurethane foam sheet over the front and bridging region of the shell and at least part of the back side, and (iii) subjecting the shell to curing/vulcanisation so that the foam becomes integrally bonded to the silicone surface. Further steps include sealing the implant through additional silicone application and curing.
In the UPC proceedings, Silimed asserts infringement of process Claims 1–19 and related product-by-process Claims 20 and 21. The claimant seeks a pan-European cease-and-desist order, declaration of infringement, information and accounting, recall and removal of infringing products from distribution channels, destruction or surrender of infringing goods, publication of the decision, and recurring penalty payments.
The Hamburg action is directed not only against Polytech as manufacturer but also against a group of its local distributors in Austria, Belgium, the Netherlands, Portugal, the Czech Republic, Spain, Switzerland, the United Kingdom and Turkey. Silimed argues that the defendants operate within a vertically integrated distribution structure and are jointly responsible for manufacture, export and commercialisation of the allegedly infringing implants.
The UPC infringement action follows a long-running ownership dispute over EP’193.
In November 2024, the Higher Regional Court Frankfurt am Main held that Polytech had filed the patent as an unauthorised applicant and ordered transfer of the patent to Silimed. The court concluded that the patented subject matter originated from Silimed within the framework of a prior contractual cooperation between the companies. The decision became final in December 2025 after the Federal Court of Justice (X ZR 111/24) rejected Polytech’s appeal against denial of leave to appeal.
The dispute dates back to a distribution relationship in the 1990s and 2000s, when Polytech acted as distributor of Silimed’s polyurethane-coated implants in Europe. After the cooperation ended in 2008, Polytech began manufacturing its own polyurethane-coated implants and filed EP’193 in 2011. Silimed initiated entitlement proceedings in 2017, alleging misuse of confidential manufacturing know-how disclosed during the cooperation.
Silimed is relying on its long-standing Brazilian counsels Karlo Tinoco and Roberto Rodrigues of RNA Law to coordinate the international enforcement campaign. They are working closely with Mike Gruber of Carpmaels & Ransford, who handled the successful entitlement proceedings in Frankfurt, together with John Brunner of Carpmaels & Ransford, and Dr Gisbert Hohagen and Michael Washbrook of Taylor Wessing, who are cooperating as UPC representatives in the infringement action.
Also reported at: https://mp.weixin.qq.com/s/vIEkOAO5mKKiUXz-vqvikQ

This section gives quick answers to the most common questions about this insight. What changed, why it matters, and the practical next steps. If your situation needs tailored advice, contact the RNA Law team.
Q1: Who filed the UPC action in Hamburg?
A1: Brazilian medical device manufacturer Silimed Indústria de Implantes Ltda filed the infringement action before the UPC, Hamburg Local Division.
Q2: Which patent is at issue in the UPC case?
A2: The case concerns European Patent EP 2 581 193 B1 (EP’193), protecting a process for manufacturing polyurethane-coated silicone implants.
Q3: What types of claims does Silimed allege are infringed?
A3: Silimed asserts infringement of process Claims 1–19 and related product-by-process Claims 20 and 21.
Q4: What remedies is Silimed seeking in the UPC proceedings?
A4: Silimed seeks a pan-European cease-and-desist order, declaration of infringement, information and accounting, recall and removal from distribution channels, destruction or surrender of goods, publication of the decision, and recurring penalty payments.
Q5: Who are the defendants besides Polytech?
A5: The action also targets several alleged local distributors in Austria, Belgium, the Netherlands, Portugal, the Czech Republic, Spain, Switzerland, the United Kingdom and Turkey.
Q6: What prior court decision is mentioned as background to the UPC action?
A6: The Higher Regional Court Frankfurt am Main ordered transfer of EP’193 to Silimed in November 2024, and the decision became final in December 2025 after the Federal Court of Justice rejected Polytech’s challenge.